
CMO Speaks is a blog featuring the voices of SCDAA’s clinical leadership team. Part 1 of our Gene Therapy series was written by Dr. Lewis Hsu and Dr. Sri Lakshmi Jamalapur.
How can you cure an inherited disease like sickle cell disease? One way would be to develop some extremely effective medications that would alleviate all the symptoms. Another would be to transplant stem cells that produce red blood cells. But the ultimate way to cure genetic disease is with gene therapy.
Gene therapy is a relatively new treatment for sickle cell warriors. Researchers began conducting experimental studies (clinical research) in people about seven years ago. At first, the only approach to this treatment was a process called “gene addition” developed by the Boston-based pharmaceutical company bluebird bio. Soon, several other groups joined with other approaches for gene therapy. By early 2022, there were six competing organizations enrolling in gene therapy clinical research. They are listed in the table below.
There has been a lot of news recently regarding gene therapy. Earlier this year, The New York Times published this article about gene therapy and sickle cell disease, which highlighted the voices and experiences of advocates from around our community and brought renewed attention to this pioneering treatment. Two pharmaceutical groups say that they will bring their gene therapy clinical research results to the U.S. Food and Drug Administration (FDA) for possible approval as safe and effective in the first quarter of 2023 (highlighted green in the table).
At the same time, in late February, three other groups announced that they are closing their gene therapy clinical research studies (highlighted yellow in the table). Some people worry that seeing companies end their gene therapy studies is not good news for sickle cell disease. However, another way to look at these announcements is that the most fruitful studies are heading toward possible FDA approval. Less productive clinical research efforts are cutting their losses and being pruned away.
It’s like a gardener doing work in a little garden to keep the plants that are bearing fruit (the two groups that will bring their results to U.S. FDA) alive but removing those plants that have stopped growing (the groups that are closing their gene therapy clinical research). The wisdom of pruning down the less productive activity is recognized by many proverbs, including “if you run after two hares, you will catch neither,” or “have too many irons in the fire,” or “he who tries to sit on two chairs will end up sitting on neither,” or John 15:1 in the Bible. Drop-out studies are part of the healthy competitive process of research and investment.
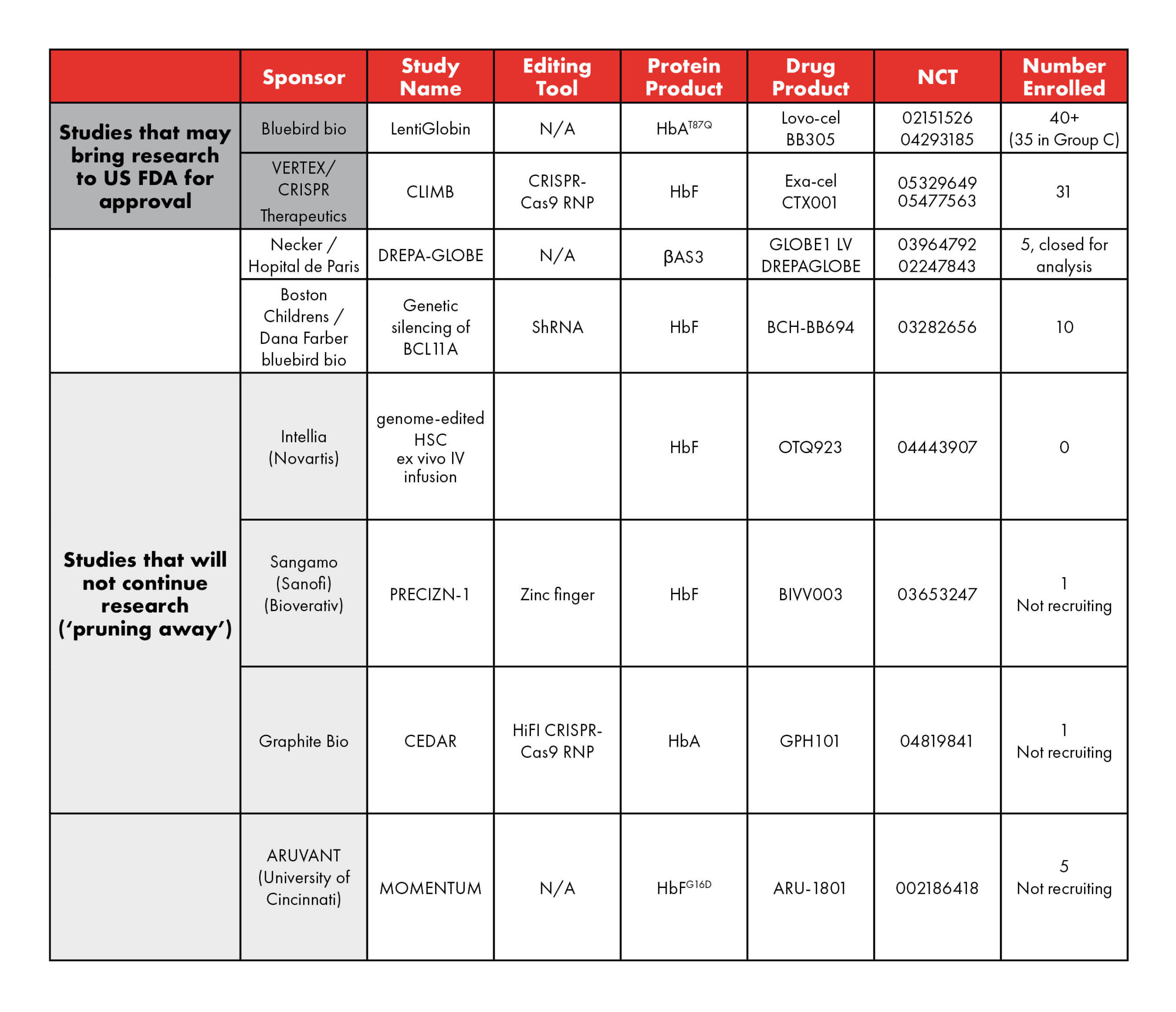
Table 1: Current and upcoming studies of gene therapy in SCD. (adapted from Kanter and Falcon 2021; Hsu and Jamalapur 2022) N/A – not applicable; LVV – lentiviral vector; HbAT87Q – Hb with a single mutation conferring most of the antisickling effect of γ-globin; βAS3 – an antisickling β-globin containing 3 amino acid substitutions in the wild-type HBB; HbFG16D – modified HbF that increases affinity to α-globin to outcompete sickle mutated β-globin; AAV6 – Adeno-Associated Virus serotype 6.
Outside of gene therapy studies, promising sickle cell progress is everywhere. Researchers are developing innovative medications and studying stem cell transplants that could make a world of difference to warriors who are unable or unwilling to undergo gene therapy in the future. The clinical research process is fine-tuned to determine which treatments are safe and effective. Sickle cell warriors are encouraged to help push this research forward by participating in clinical trials.
Lewis Hsu, MD, PhD, is a pediatric hematologist who serves as director of the Sickle Cell Center and professor of pediatrics for the University of Illinois at Chicago. He has conducted sickle cell research, published over 50 peer-reviewed papers and co-authored “Hope and Destiny: The Patient and Parent’s Guide to Sickle Cell Disease and Sickle Cell Trait.” He currently serves as the SCDAA Chief Medical Officer.
Sri Lakshmi Jamalapur, MD, is a pediatric hematologist and the new leader for the Pediatric Sickle Cell Center at Children’s Hospital of Michigan in Detroit. She has collaborated with Dr. Hsu on health education and sickle cell advocacy since 2019.





